This web page was produced as an assignment for Genetics 564, an undergraduate course at UW-Madison.
Cri du Chat Syndrome
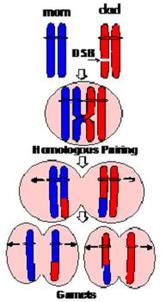
Cri du Chat syndrome is French for "cry of the cat." It is a rare genetic disease with an estimated prevalence of 1 in 15,000 to 50,000 live-born infants [1]. It is caused by a deletion that varies in size in the short arm of chromosome 5 [1]. Typically, the deletion occurs from unequal recombination or chromosomal breakage during meiosis when the sperm or egg is forming (refer to Figure 1). If the affected gamete is fertilized, the child will have Cri du Chat. Because the break in the chromosome often happens in the gamete cell (sperm or egg), the parent doesn't necessarily have to have the break in other cells or have the syndrome themselves for the child to develop Cri du Chat. Females are more commonly affected than males [2]. The chromosome with the deletion is inherited from the father's sperm in approximately 80 percent of the cases [3].
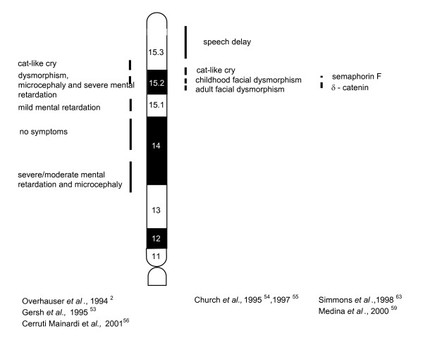
Due to the variation in the deletion of chromosome 5, there is phenotypic (observable characteristics) and cytogenetic (structure and function of chromosomes) inconsistency in severity amongst the patients diagnosed with Cri du Chat. The variation indicates that there is correlation between the clinical severity in symptoms and the size or type of deletion [4]. The telomerase reverse transcripatase (TERT) gene and the semaphorin 5A (SEMA5A) gene are often deleted from chromosome 5. (The TERT gene helps keep the telomeres intact during cell division and the SEMA5A gene functions in neural development) [3]. The recognizable phenotype associated with the deletion of these genes indicates that a critical region is involved in causing the characteristic clinical symptoms (refer to Figure 2 for clinical symptoms and critical regions) [1].
|
Figure 1. DNA recombination and repair. When the chromosomes are lined up at the metaphase, there is the possibility of recombination. If the chromosome pairs do not line up correctly or if the chromosome has breaks that are not repaired, deletion or replication, which can cause miscarriages or chromosomal disorders, can occur [5].
|
Figure 2. Phenotypic map of critical regions. Vertical lines indicate the critical regions in the short arm of chromosome 5. Deletions of these critical regions can cause the various Cri du Chat symptoms.
|
Symptoms
The most well known symptom of Cri du Chat is the high-pitched, cat-like cry, which can be heard in Video 1. The abnormal cry is caused by abnormal development of tissues in the larynx (hollow muscular organ in air passage) and epiglottis (flap of cartilage at the entrance of the larynx).
Video 1. Youtube 5.11.2011. Gid- Cri Du Chat. Retrieved from here.
Described characteristics of this syndrome include: small birth weights, microcephaly (abnormal smallness of head), poor muscle tone, cognitive impairment, and respiratory problems. Those with Cri du Chat also have well described dsymorphic features including: a round face, small chin, wide-set eyes, folds of skin over their eyes, a small nasal bridge, low set ears, micrognathia (small lower jaw), partial webbing of the fingers and/or toes, and a single palmar crease [2][3].
Individuals with Cri du Chat can also have internal abnormalities, such as heart, muscular, or skeletal defects and hearing or sight problems. Because of the muscular defects, when these individuals grow older, they can have difficulty with mobility and communication. Occasionally, these individuals have behavioral problems but they typically have a gentle and affectionate personality overall [1][3].
If no critical medical conditions or organ defects exist, life expectancy for those with Cri du Chat is normal [3].
Individuals with Cri du Chat can also have internal abnormalities, such as heart, muscular, or skeletal defects and hearing or sight problems. Because of the muscular defects, when these individuals grow older, they can have difficulty with mobility and communication. Occasionally, these individuals have behavioral problems but they typically have a gentle and affectionate personality overall [1][3].
If no critical medical conditions or organ defects exist, life expectancy for those with Cri du Chat is normal [3].
SEMA5A Gene
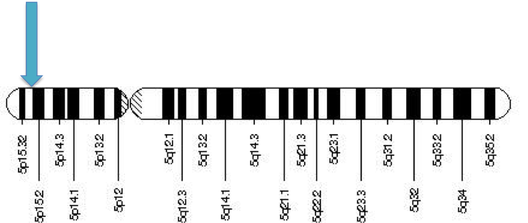
The SEMA5A gene is fundamental for encoding membrane proteins for proper axonal guidance during neural development [6]. SEMA5A has 26 exons and is located on the short arm of fifth chromosome (map location 5p15.31) [accession number: XM_006714506.1, FASTA] [7]. The gene has 11,946 base pairs that code for a 1,074-amino acid protein [accession number: NP_003957.2, FASTA] [8].
Figure 3. Human chromosome 5. The blue arrow indicates the region of the SEMA5A gene (5p15.31).
SEMA5A is part of the semaphorin gene family, which encodes for semaphorin domain membrane proteins (responsible for protein binding) and multiple thrombospondin type-1 repeats (responsible for regulation of cellular interactions in vertebrates) [6]. Proteins from the semaphorin gene family are cysteine-rich and are secreted or used as transmembrane proteins (proteins that span from one side to the other side of the membrane). Semaphorin proteins are unique to animals tissues and are especially prevalent in nervous tissue [9].
SEMA5A works by using a bifunctional guidance cue, which exhibits both alluring and inhibitory stimuli on the fasciculus retroflexus. Fasciculus retroflexus is a diencephalon (posterior part of the brain) fiber involved in limbic function. The effects of the SEMA5A cues on neural activity are reliant upon the bound proteoglycan (protein bound to glycosaminoglycan groups). There is an alluring cue when attached to heparan sulfate proteoglycans (a linear polysaccharide chain found in all animal tissue) and an inhibitory cue when attached to chondroitin sulfate proteoglycans (a protein bound to a chondroitin sulfate side chain) [10].
In the instance that SEMA5A is deleted, such as what is seen in Cri du Chat syndrome patients, proper neuronal development does not occur. This is because the deletion of the SEMA5A gene leads to an inadequate amount of the SEMA5A protein, which provides the bifunctional guidance cues that were previously described. Without these cues, the axonal guidance during neuronal development would be inhibited, leading to the cognitive impairment seen in Cri du Chat syndrome patients.
The deletion of SEMA5A has been linked to autism, Parkinson's disease, mental retardation, and Cri du Chat [7]. The SEMA5A gene contains approximately 10% of the Cri du Chat critical region [8].
SEMA5A works by using a bifunctional guidance cue, which exhibits both alluring and inhibitory stimuli on the fasciculus retroflexus. Fasciculus retroflexus is a diencephalon (posterior part of the brain) fiber involved in limbic function. The effects of the SEMA5A cues on neural activity are reliant upon the bound proteoglycan (protein bound to glycosaminoglycan groups). There is an alluring cue when attached to heparan sulfate proteoglycans (a linear polysaccharide chain found in all animal tissue) and an inhibitory cue when attached to chondroitin sulfate proteoglycans (a protein bound to a chondroitin sulfate side chain) [10].
In the instance that SEMA5A is deleted, such as what is seen in Cri du Chat syndrome patients, proper neuronal development does not occur. This is because the deletion of the SEMA5A gene leads to an inadequate amount of the SEMA5A protein, which provides the bifunctional guidance cues that were previously described. Without these cues, the axonal guidance during neuronal development would be inhibited, leading to the cognitive impairment seen in Cri du Chat syndrome patients.
The deletion of SEMA5A has been linked to autism, Parkinson's disease, mental retardation, and Cri du Chat [7]. The SEMA5A gene contains approximately 10% of the Cri du Chat critical region [8].
Diagnosis and Treatment
Often, clinicians are initially alerted to the possibility of the diagnosis of Cri du Chat by the distinctive cry of the babies affected, which then leads to further, more detailed, morphologic evaluation and possible genetic specialist referral. However, there are a few methods utilized to detect chromosomal abnormalities before the child is ever born.
One method of prenatal testing is called chorionic villus sampling. This method involves taking a sample from outside the amniotic sac, through the mother's abdominal and uterine walls or through a small tube inserted into the vagina. Testing can be done within the first trimester and the results are received in two weeks or less. There is a 1-3 percent risk of miscarriage following the procedure. The results are less accurate than amniocentesis, but they are about 98 percent accurate in diagnosing Down Syndrome and some other chromosomal abnormalities [11].
Another method of prenatal genetic testing is called an amniocentesis, which is a procedure that involves directly obtaining amniotic fluid from the mother, with a needle, under ultrasound guidance. Testing isn't done until the second trimester to improve accuracy and avoid damage to the baby. There is a 0.3-0.5 percent risk of miscarriage following the procedure. The results are received 3 to 4 weeks following the test and they typically have 99 percent accuracy in determining most chromosomal diseases [11].
Additionally, there is now a noninvasive prenatal testing option. It is a method of screening that involves a maternal blood test to investigate the fetal DNA for sex, blood type, and risk of chromosomal abnormalities. This method of testing is only available for women who are high risk for chromosomal conditions. Noninvasive prenatal testing poses no risk of miscarriage due to the procedure, although it has lower specificity and sensitivity than the other methods. The results are typically received two weeks following the procedure [12].
Although there is no direct specific therapy for Cri du Chat, speech therapy, occupational therapy, and physical therapy can help improve language and motor skills.
One method of prenatal testing is called chorionic villus sampling. This method involves taking a sample from outside the amniotic sac, through the mother's abdominal and uterine walls or through a small tube inserted into the vagina. Testing can be done within the first trimester and the results are received in two weeks or less. There is a 1-3 percent risk of miscarriage following the procedure. The results are less accurate than amniocentesis, but they are about 98 percent accurate in diagnosing Down Syndrome and some other chromosomal abnormalities [11].
Another method of prenatal genetic testing is called an amniocentesis, which is a procedure that involves directly obtaining amniotic fluid from the mother, with a needle, under ultrasound guidance. Testing isn't done until the second trimester to improve accuracy and avoid damage to the baby. There is a 0.3-0.5 percent risk of miscarriage following the procedure. The results are received 3 to 4 weeks following the test and they typically have 99 percent accuracy in determining most chromosomal diseases [11].
Additionally, there is now a noninvasive prenatal testing option. It is a method of screening that involves a maternal blood test to investigate the fetal DNA for sex, blood type, and risk of chromosomal abnormalities. This method of testing is only available for women who are high risk for chromosomal conditions. Noninvasive prenatal testing poses no risk of miscarriage due to the procedure, although it has lower specificity and sensitivity than the other methods. The results are typically received two weeks following the procedure [12].
Although there is no direct specific therapy for Cri du Chat, speech therapy, occupational therapy, and physical therapy can help improve language and motor skills.
References
Youtube video: https://www.youtube.com/watch?v=TYQrzFABQHQ
Figure 1. Manning, K. (1977). The larynx in the Cri du Chat Syndrome. The Journal of Laryngology & Otology, 91(10), 887-892.
Figure 2. Cerruti Mainardi, P. (2006). Cri du Chat syndrome. Orphanet Journal of Rare Diseases, 1, 33. doi:10.1186/1750-1172-1-33.
Figure 3. Chromosome 5. (2013, January 27). Retrieved January 30, 2015, from http://ghr.nlm.nih.gov/chromosome/5
[1] Cerruti Mainardi, P. (2006). Cri du Chat syndrome. Orphanet Journal of Rare Diseases, 1, 33. doi:10.1186/1750-1172-1-33.
[2] Manning, K. (1977). The larynx in the Cri du Chat Syndrome. The Journal of Laryngology & Otology, 91(10), 887-892.
[3] Genetic Science Learning Center (2014, June 22) Cri-du-Chat Syndrome. Learn.Genetics. Retrieved January 28, 2015, from http://learn.genetics.utah.edu/content/disorders/chromosomal/cdc/
[4] Mainardi, P., Perfumo, C., Cali, A., Coucourde, G., Pastore, G., Cavani, S., Bricarelli, F. (2001). Clinical and molecular characterization of 80 patients with 5p deletion: genotype-phenotype correlation. Journal of Medical Genetics,38(3), 151–158. doi:10.1136/jmg.38.3.151.
[5] Manning, K. (1977). The larynx in the Cri du Chat Syndrome. The Journal of Laryngology & Otology, 91(10), 887-892.
[6] Genes and mapped phenotypes. (2015, January 13). Retrieved January 30, 2015, from http://www.ncbi.nlm.nih.gov/gene/9037
[7] Chen, C., Huang, M., Chen, Y., Chern, S., Wu, P., Su, J., Wang, W. (2013). Cri-du-chat (5p-) syndrome presenting with cerebellar hypoplasia and hypospadias: Prenatal diagnosis and aCGH characterization using uncultured amniocytes. Gene, 524(2), 407-411.
[8] Hamosh, A. (2005, April 1). OMIM Entry - * 609297 - SEMAPHORIN 5A; SEMA5A. Retrieved January 30, 2015, from http://www.omim.org/entry/609297
[9] Yazdani, U., & Terman, J. (2006, March 30). The semaphorins. Retrieved January 30, 2015, from http://genomebiology.com/2006/7/3/211
[10] Kantor, D., Chivatakarn, O., Peer, K., Oster, S., Inatani, M., Hansen, M., Kolodkin, A. (2004). Semaphorin 5A Is a Bifunctional Axon Guidance Cue Regulated by Heparan and Chondroitin Sulfate Proteoglycans. Neuron,44(6), 961-975.
[11] O'Neil, D. (2012, January 1). Human Chromosomal Abnormalities: Detection. Retrieved January 28, 2015, from http://anthro.palomar.edu/abnormal/abnormal_2.htm
[12] Mayo Clinic Staff. (2013, February 23). Noninvasive prenatal testing. Retrieved February 3, 2015, from http://www.mayoclinic.org/tests-procedures/noninvasive-prenatal-testing/basics/definition/prc-20012964
Figure 1. Manning, K. (1977). The larynx in the Cri du Chat Syndrome. The Journal of Laryngology & Otology, 91(10), 887-892.
Figure 2. Cerruti Mainardi, P. (2006). Cri du Chat syndrome. Orphanet Journal of Rare Diseases, 1, 33. doi:10.1186/1750-1172-1-33.
Figure 3. Chromosome 5. (2013, January 27). Retrieved January 30, 2015, from http://ghr.nlm.nih.gov/chromosome/5
[1] Cerruti Mainardi, P. (2006). Cri du Chat syndrome. Orphanet Journal of Rare Diseases, 1, 33. doi:10.1186/1750-1172-1-33.
[2] Manning, K. (1977). The larynx in the Cri du Chat Syndrome. The Journal of Laryngology & Otology, 91(10), 887-892.
[3] Genetic Science Learning Center (2014, June 22) Cri-du-Chat Syndrome. Learn.Genetics. Retrieved January 28, 2015, from http://learn.genetics.utah.edu/content/disorders/chromosomal/cdc/
[4] Mainardi, P., Perfumo, C., Cali, A., Coucourde, G., Pastore, G., Cavani, S., Bricarelli, F. (2001). Clinical and molecular characterization of 80 patients with 5p deletion: genotype-phenotype correlation. Journal of Medical Genetics,38(3), 151–158. doi:10.1136/jmg.38.3.151.
[5] Manning, K. (1977). The larynx in the Cri du Chat Syndrome. The Journal of Laryngology & Otology, 91(10), 887-892.
[6] Genes and mapped phenotypes. (2015, January 13). Retrieved January 30, 2015, from http://www.ncbi.nlm.nih.gov/gene/9037
[7] Chen, C., Huang, M., Chen, Y., Chern, S., Wu, P., Su, J., Wang, W. (2013). Cri-du-chat (5p-) syndrome presenting with cerebellar hypoplasia and hypospadias: Prenatal diagnosis and aCGH characterization using uncultured amniocytes. Gene, 524(2), 407-411.
[8] Hamosh, A. (2005, April 1). OMIM Entry - * 609297 - SEMAPHORIN 5A; SEMA5A. Retrieved January 30, 2015, from http://www.omim.org/entry/609297
[9] Yazdani, U., & Terman, J. (2006, March 30). The semaphorins. Retrieved January 30, 2015, from http://genomebiology.com/2006/7/3/211
[10] Kantor, D., Chivatakarn, O., Peer, K., Oster, S., Inatani, M., Hansen, M., Kolodkin, A. (2004). Semaphorin 5A Is a Bifunctional Axon Guidance Cue Regulated by Heparan and Chondroitin Sulfate Proteoglycans. Neuron,44(6), 961-975.
[11] O'Neil, D. (2012, January 1). Human Chromosomal Abnormalities: Detection. Retrieved January 28, 2015, from http://anthro.palomar.edu/abnormal/abnormal_2.htm
[12] Mayo Clinic Staff. (2013, February 23). Noninvasive prenatal testing. Retrieved February 3, 2015, from http://www.mayoclinic.org/tests-procedures/noninvasive-prenatal-testing/basics/definition/prc-20012964